Introduction:
A single nucleotide polymorphism in the promoter region of the Duffy antigen/chemokine receptor (DARC) gene results in the red blood cell (RBC) Duffy-null phenotype, which is present in up to 80% of individuals of Sub-Saharan ancestry. Duffy-null phenotype is associated with lower peripheral absolute neutrophil counts (ANC) with preserved myeloid maturation and neutrophil function. The impact of Duffy-null phenotype on patients with hematologic malignancies is largely unknown; however, lower ANC (both at baseline and during treatment) may lead to the receipt of less intense treatment (with lower upfront dosing as well as more treatment interruptions/dose reductions). Lower ANC also pose challenges in enrollment in clinical trials due to ANC cutoffs for eligibility criteria. Herein, we evaluated the impact of Duffy phenotype status on the incidence of neutropenic fever and chemotherapy dose reductions in a cohort of patients treated for multiple myeloma (MM) or light chain (AL) amyloidosis at the largest safety net hospital in New England, USA.
Methods:
In this retrospective study, we identified patients treated for MM or AL amyloidosis at Boston University Medical Center (BMC) who underwent phenotyping for the RBC Duffy antigen. Patients were categorized as Duffy-null phenotype, i.e., Fy (a-b-) or non-Duffy-null phenotype including Duffy heterozygous, i.e., Fy (a-b+ or a+b-) or Duffy positive, i.e., Fy (a+b+). We collected data from medical charts including patient demographics; clinical characteristics (including malignancy and treatment regimens); incidence of neutropenia (defined as ANC <1,800/µL); incidence of neutropenic fever (excluding peri-stem cell transplantation [SCT] neutropenia); and chemotherapy dose adjustments. This research was approved by the BMC Institutional Review Board.
Results:
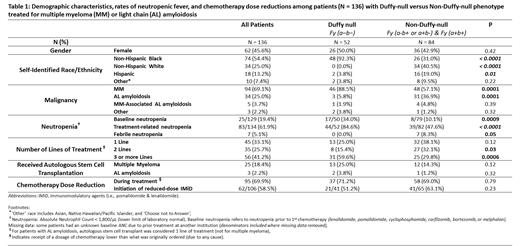
Between 06/2020 and 06/2023, 136 patients had a Duffy phenotype evaluation; mean age at the time of first chemotherapy was 65 (± 11.3) years and 62/136 (46%) were female. The most common self-identified race/ethnicity was non-Hispanic Black (74/136, 54%), followed by non-Hispanic White (34/136, 25%), Hispanic (18/136, 13%), and other (10/36, 7%), and other (10/136, 6%). Plasma cell dyscrasias included MM (94/136, 69%), AL amyloidosis (34/136, 25%) (Table 1).
Duffy-null Fy (a-b-) phenotype was seen in 52/136 (38%) patients, while 84/136 (62%) were non-Duffy-null, including 73/136 (54%) with Duffy heterozygosity Fy (a-b+ or a+b-) and 11/136 (8%) with Duffy positivity Fy (a+ b+). Number of lines of treatment did not differ by Duffy status (Table 1): 35/136 (26%) received 2 lines of treatment; 56/136 (41%) received ≥ 3 lines; and 28/136 (21%) received high-dose chemotherapy and autologous SCT.
Among those with known baseline ANC, we observed a higher frequency of neutropenia before receipt of any chemotherapy in patients with Duffy-null status (17/50, 34%) compared to those with non-Duffy-null status (8/79, 10%), p= 0.0009. Similarly, the incidence of treatment-related neutropenia (excluding the peri-SCT period) was higher in Duffy-null patients (44/52, 85%) compared to non-Duffy-null (39/82, 48%) patients, p<0.0001.
Overall, the incidence of febrile neutropenia in our cohort was low; all 7 patients who experienced a febrile neutropenic event were non-Duffy-null (7/84, 8%). None of the patients in the Duffy-null cohort (0/52, 0%) had febrile neutropenia, despite higher incidences of both baseline and treatment-related neutropenia.
Treatment interruptions/delays or dose reductions due to any cause were observed in 95/136 (70%) of all patients and did not differ by Duffy phenotype status (Table 1). The proportion of patients initiated on upfront reduced-dose pomalidomide or lenalidomide was 62/106 (59%) and also did not differ by Duffy phenotype status.
Conclusion:
In this retrospective study, we found that compared to patients with non-Duffy-null phenotype, patients with Duffy-null phenotype treated for plasma cell dyscrasias did not have a higher incidence of neutropenic fever, treatment interruptions or dose reductions, despite having higher frequencies of both baseline and treatment-related neutropenia. Prospective validation of our results could help to inform the creation and implementation of guidelines for the management of patients with lower ANC related to Duffy-null phenotype who undergo treatment for plasma cell dyscrasias.
Disclosures
Sanchorawala:Janssen, Alexion, Prothena, Celgene, Takeda, Abbvie, Regeneron, Pfizer, AstraZeneca: Consultancy, Honoraria, Membership on an entity's Board of Directors or advisory committees, Research Funding. Sloan:CTI Biopharma: Honoraria; Seagen: Research Funding; Servier Pharmaceuticals: Honoraria; Stemline Pharmaceuticals: Consultancy; Nuvectis Pharaceuticals: Consultancy; Abbvie: Consultancy. Szalat:Jansse Pharmaceuticals: Other: Participation to one advisory board.